Spatial patterns and environmental associations of deep scattering layers in the northwestern subtropical Pacific Ocean
-
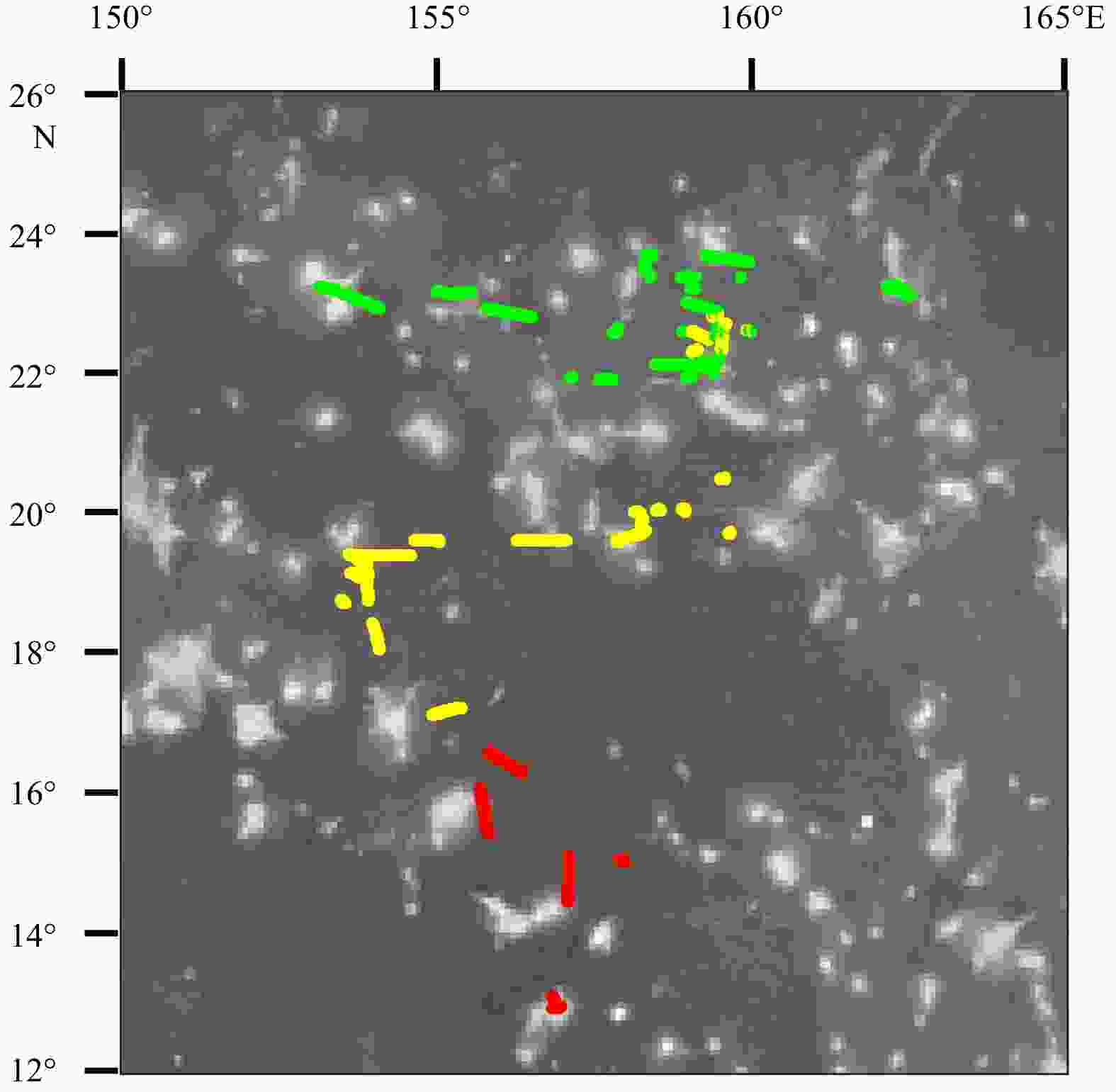
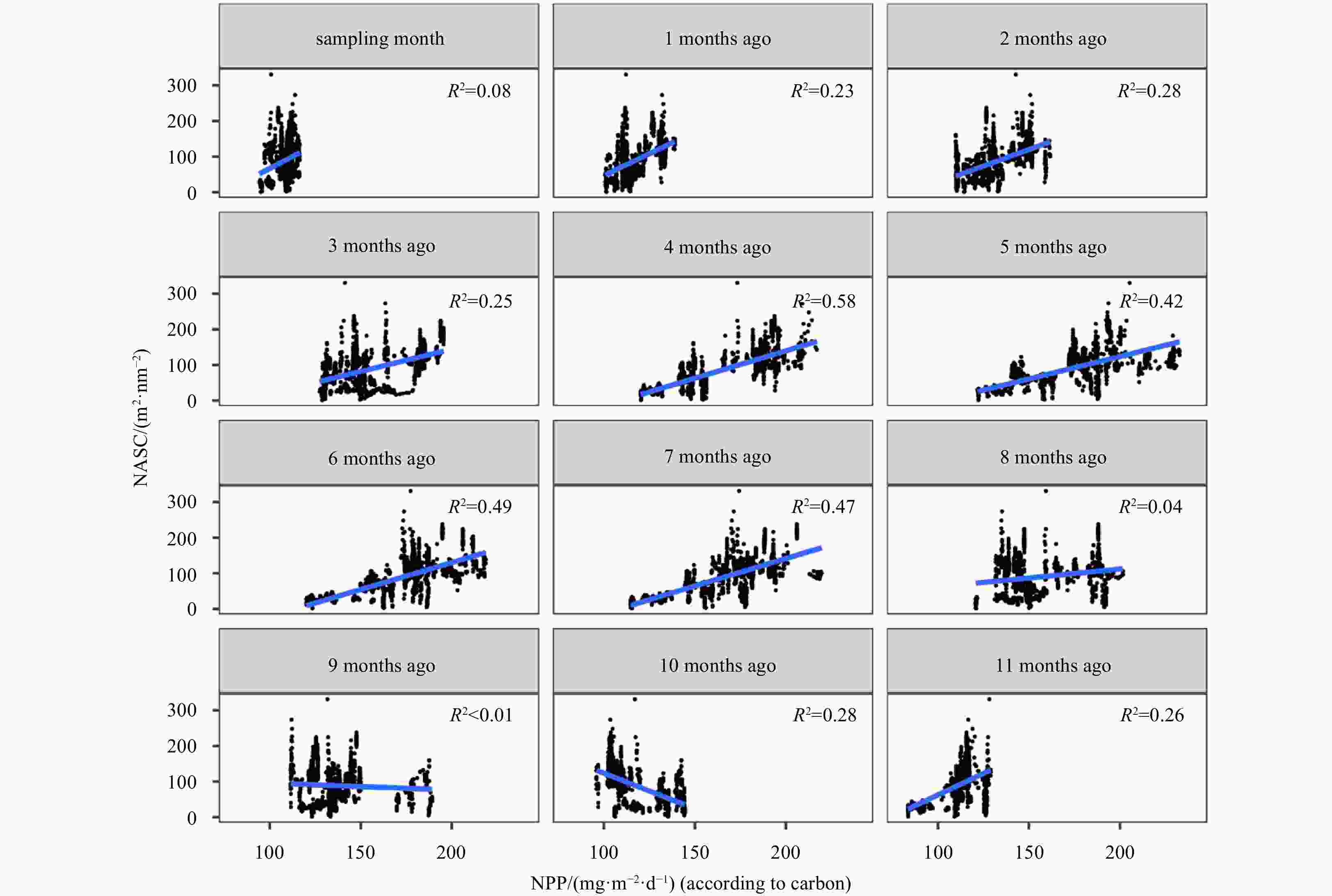
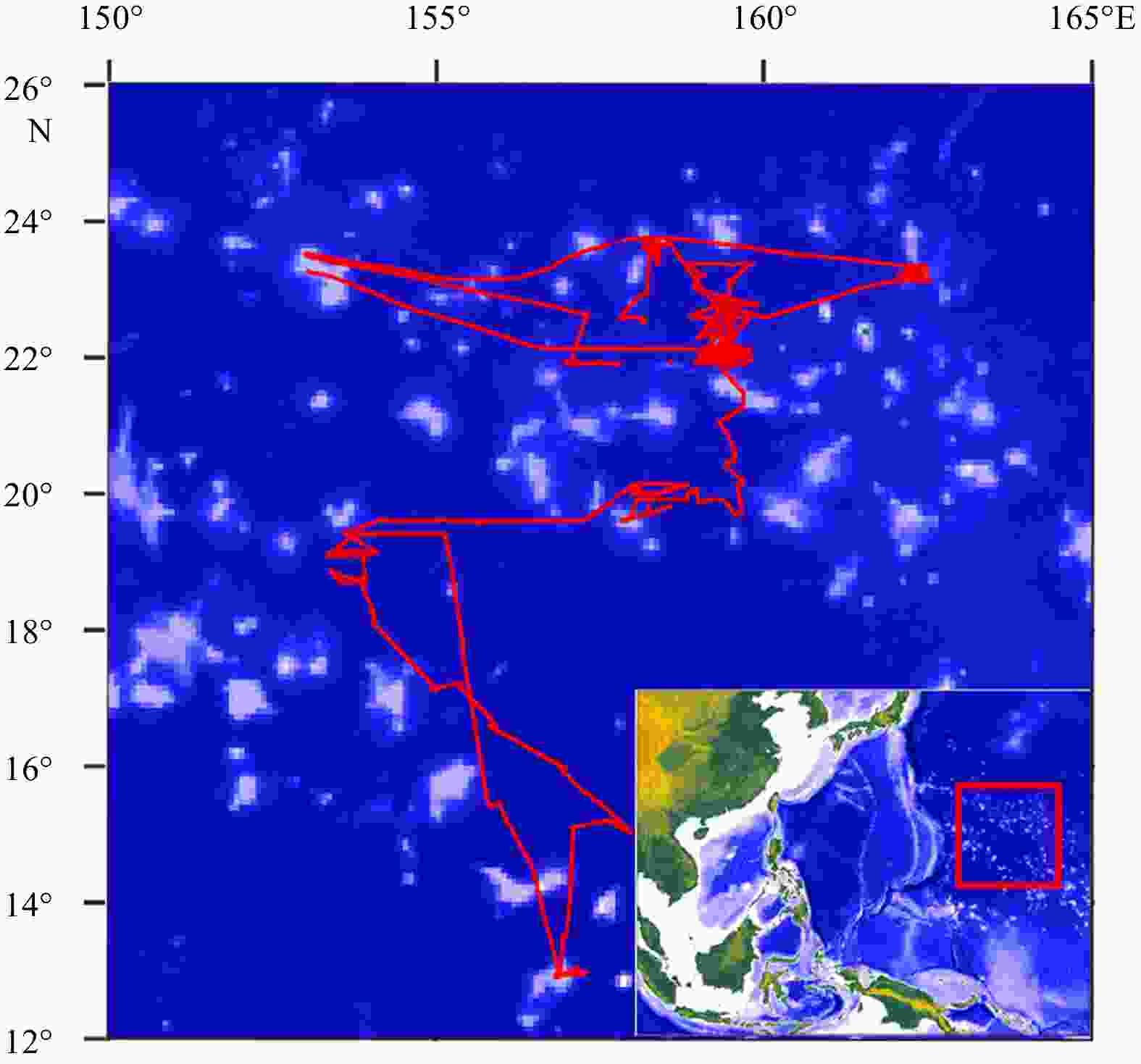
Abstract: The mesopelagic communities are important for food web and carbon pump in ocean, but the large-scale studies of them are still limited until now because of the difficulties on sampling and analyzing of mesopelagic organisms. Mesopelagic organisms, especially micronekton, can form acoustic deep scattering layers (DSLs) and DSLs are widely observed. To explore the spatial patterns of DSLs and their possible influencing factors, the DSLs during daytime (10:00–14:00) were investigated in the subtropical northwestern Pacific Ocean (13°–23.5°N, 153°–163°E) using a shipboard acoustic Doppler current profiler at 38 kHz. The study area was divided into three parts using k-means cluster analysis: the northern part (NP, 22°–24°N), the central part (CP, 17°–22°N), and the southern part (SP, 12°–17°N). The characteristics of DSLs varied widely with latitudinal gradient. Deepest core DSLs (523.5 m±17.4 m), largest nautical area scattering coefficient (NASC) (130.8 m2/n mile2±41.0 m2/n mile2), and most concentrated DSLs (mesopelagic organisms gathering level, 6.7%±0.7%) were observed in NP. The proportion of migration was also stronger in NP (39.7%) than those in other parts (18.6% in CP and 21.5% in SP) for mesopelagic organisms. The latitudinal variation of DSLs was probably caused by changes in oxygen concentration and light intensity of mesopelagic zones. A positive relationship between NASC and primary productivity was identified. A four-months lag was seemed to exist. This study provides the first basin-scale baselines information of mesopelagic communities in the northwest Pacific with acoustic approach. Further researches are suggested to gain understandings of seasonal and annual variations of DSLs in the region.
-
Key words:
- deep scattering layers /
- mesopelagic community /
- baseline /
- diel vertical migration /
- zooplankton /
- micronekton
-
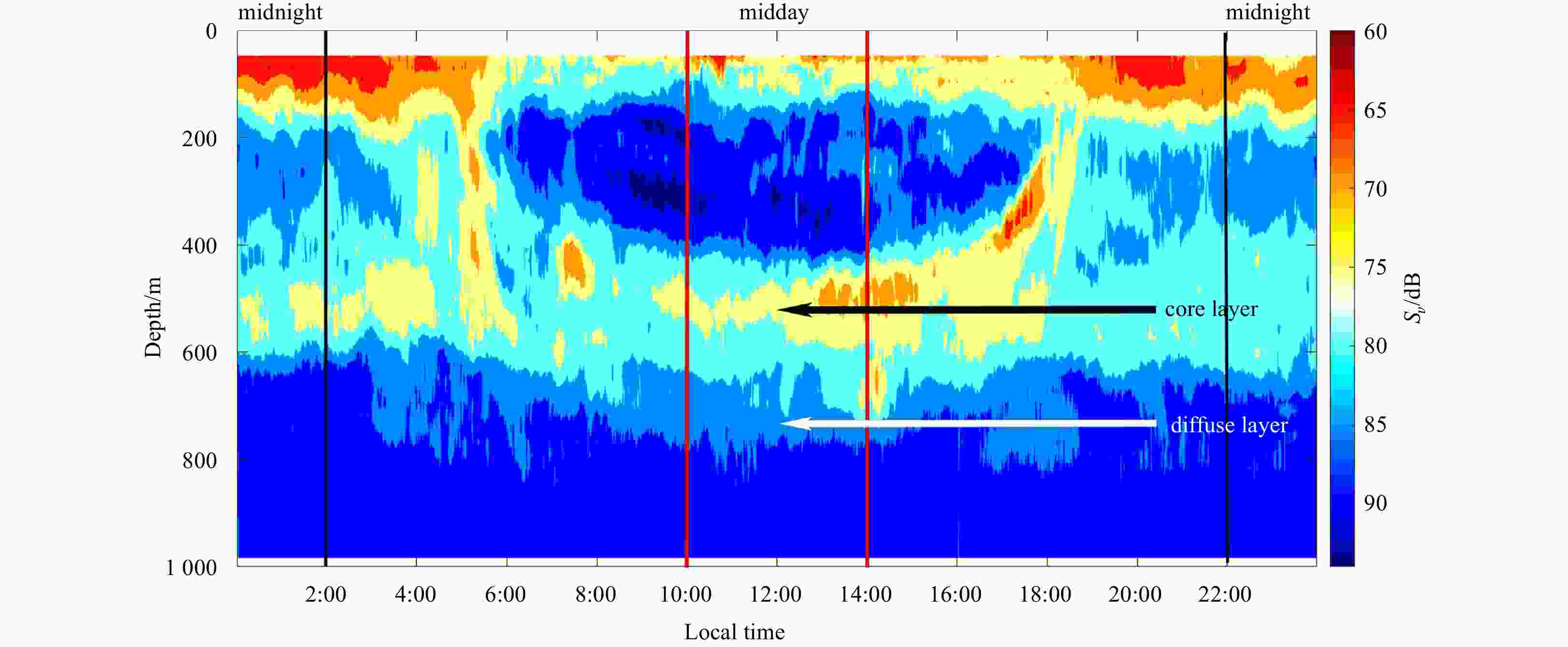
Figure 2. Echogram (38 kHz) after noise-removal showing the diel vertical migration of mesopelagic organisms (at local time 0:00–24:00, September 10, 2018). The red lines indicated the period of midday (at local time 10:00–14:00), when the dwelling depth of mesopelagic organisms was relative stable and it was easy to eliminate the disturbance of diel vertical migration. And the black lines indicated the period of midnight (at local time 22:00–next day 2:00). The black arrow indicated the core part of deep scattering layers (DSLs), and the white arrow indicated the diffuse part of DSLs.
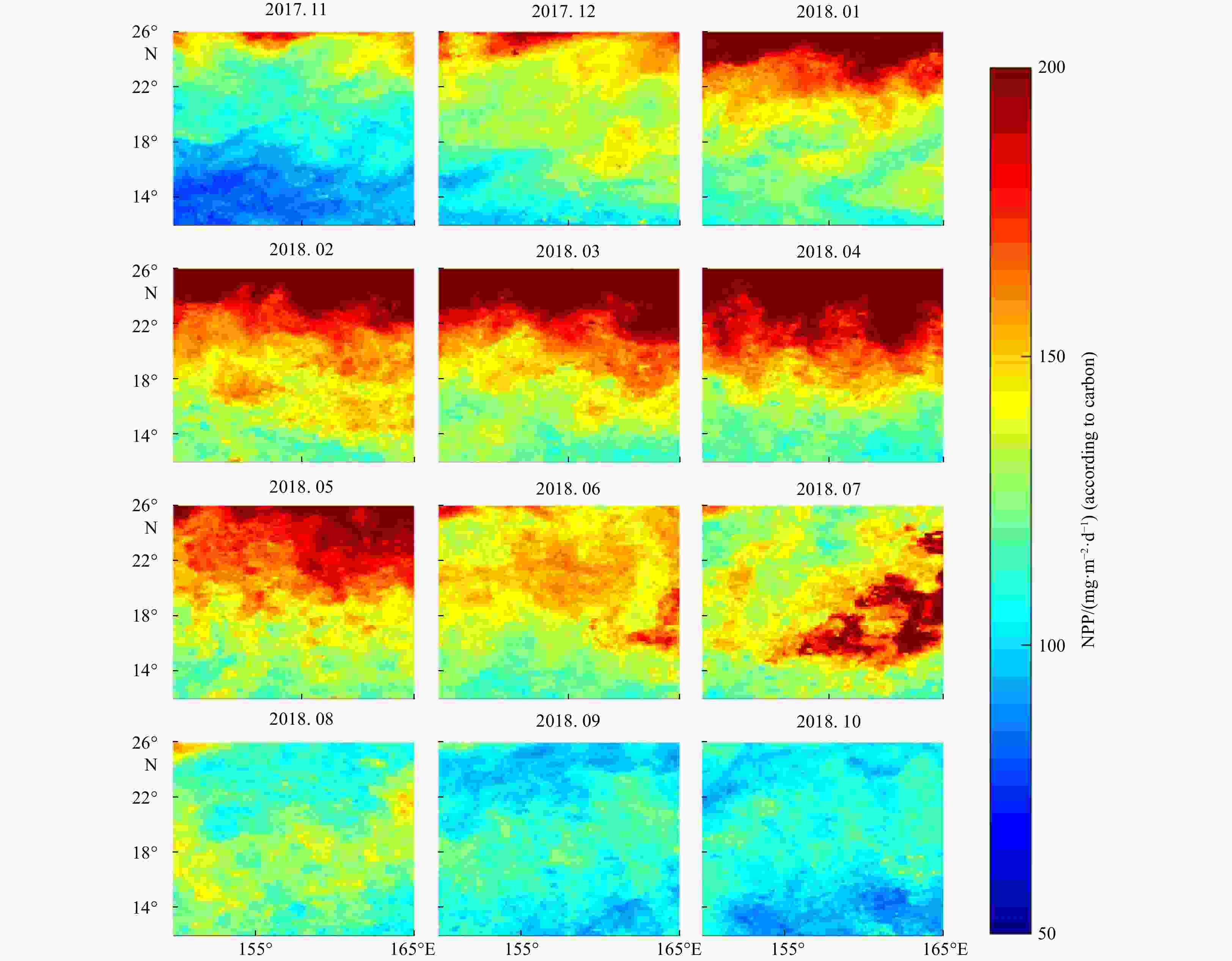
Figure 4. Maps of environmental conditions in the study area (the average data during September was used for mapping). a, b. Sea surface temperature (SST) and mesopelagic average temperature (MAT); c, d. sea surface salinity (SSS) and mesopelagic average salinity (MAS); e. mixing layer depth (MLD); f. mesopelagic average dissolved oxygen concentration (MAO); g. net primary productivity (NPP); h. 490 nm light attenuation coefficient (LAC). The black curves in some images are isolines.
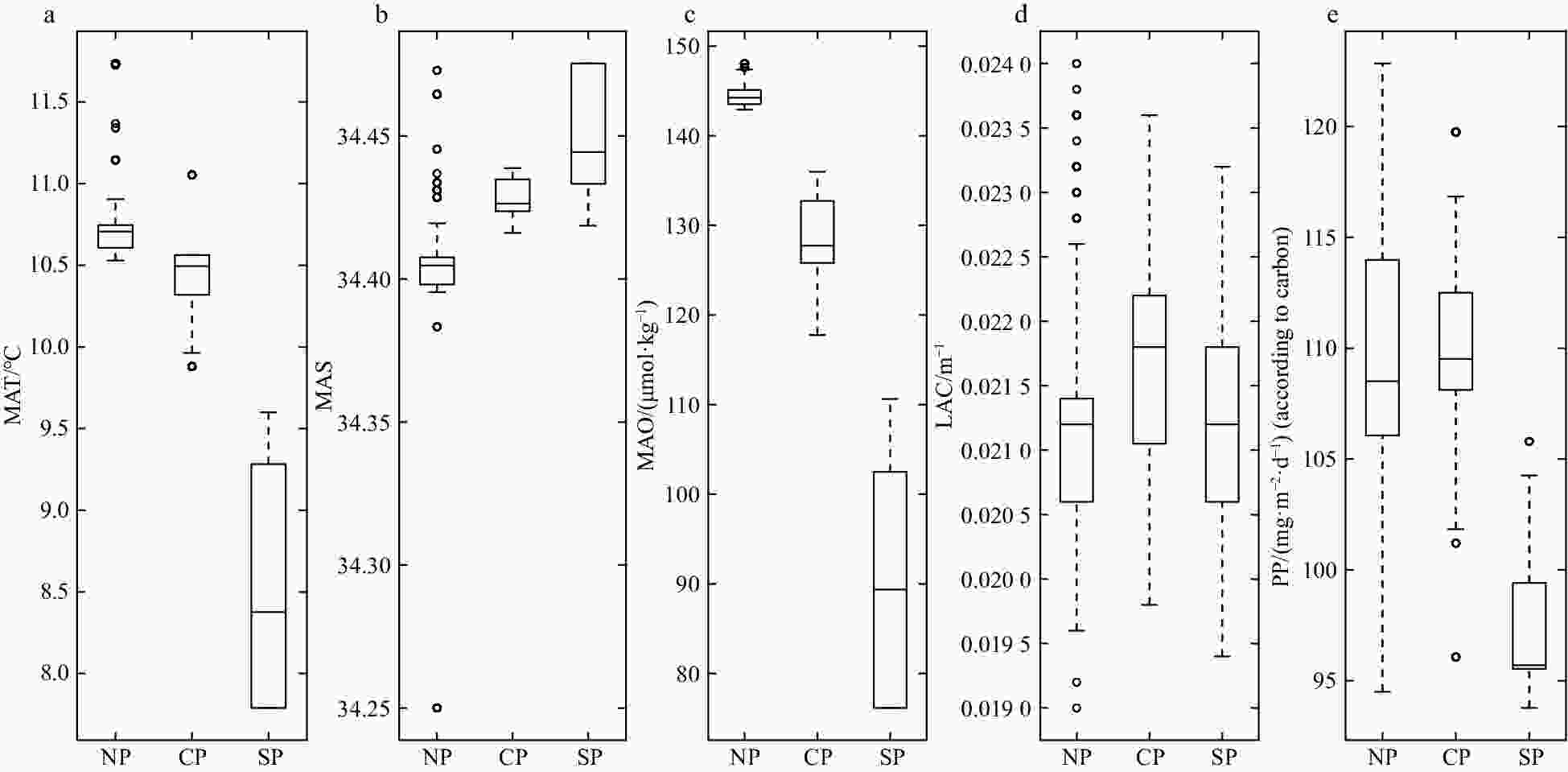
Figure 6. Box plots about the features of environment in three parts (NP, northern part; CP, central part; SP, southern part). a. Mesopelagic average temperature (MAT); b. mesopelagic average salinity (MAS); c. mesopelagic average dissolved oxygen concentration (MAO); d. 490 nm light attenuation coefficient (LAC); e. primary productivity (PP).
Figure 7. Box plots about the features of deep scattering layers (DSLs), showing the differences among three parts (NP, northern part; CP, central part; SP, southern part). a. Mesopelagic nautical area scattering coefficient (MNASC); b. center mass (CM); and c. mesopelagic gathering level (MGL) of mesopelagic zone. d and e showed the upper (UBD) and lower boundary depth (LBD) of DSLs, respectively.
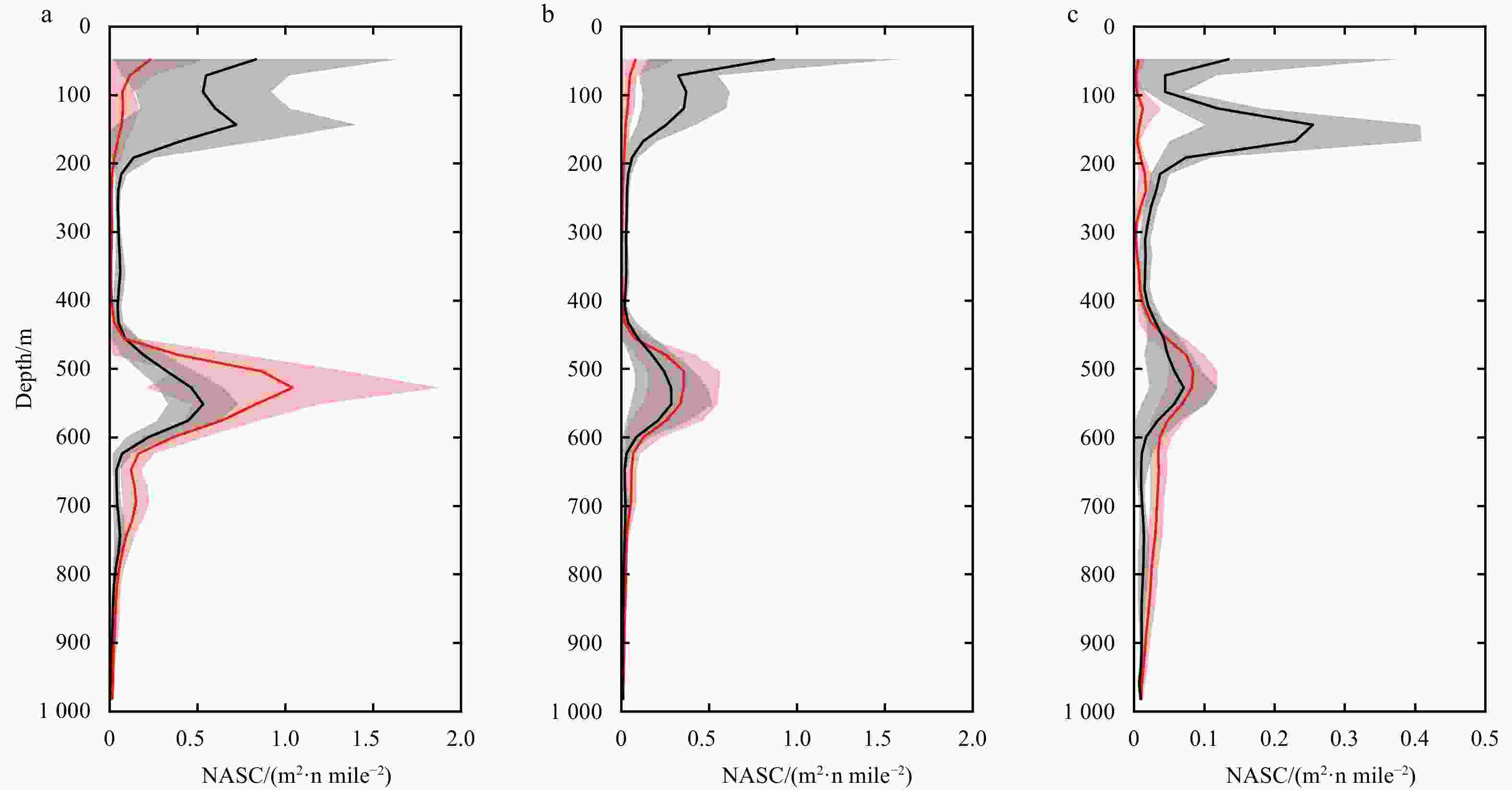
Figure 8. Vertical distribution of acoustic backscatter grouped into three parts, according to the result of k-means cluster analysis. a, northern part; b, central part; c, southern part. Red lines show average midday-time profiles (10:00–14:00), and light red shadows are range of standard deviations. Black lines show average midnight-time profiles (22:00–next day 2:00), and gray shadows are range of standard deviations. x-axis is acoustic backscatter measured as nautical area scattering coefficient (NASC) at the corresponding depth.
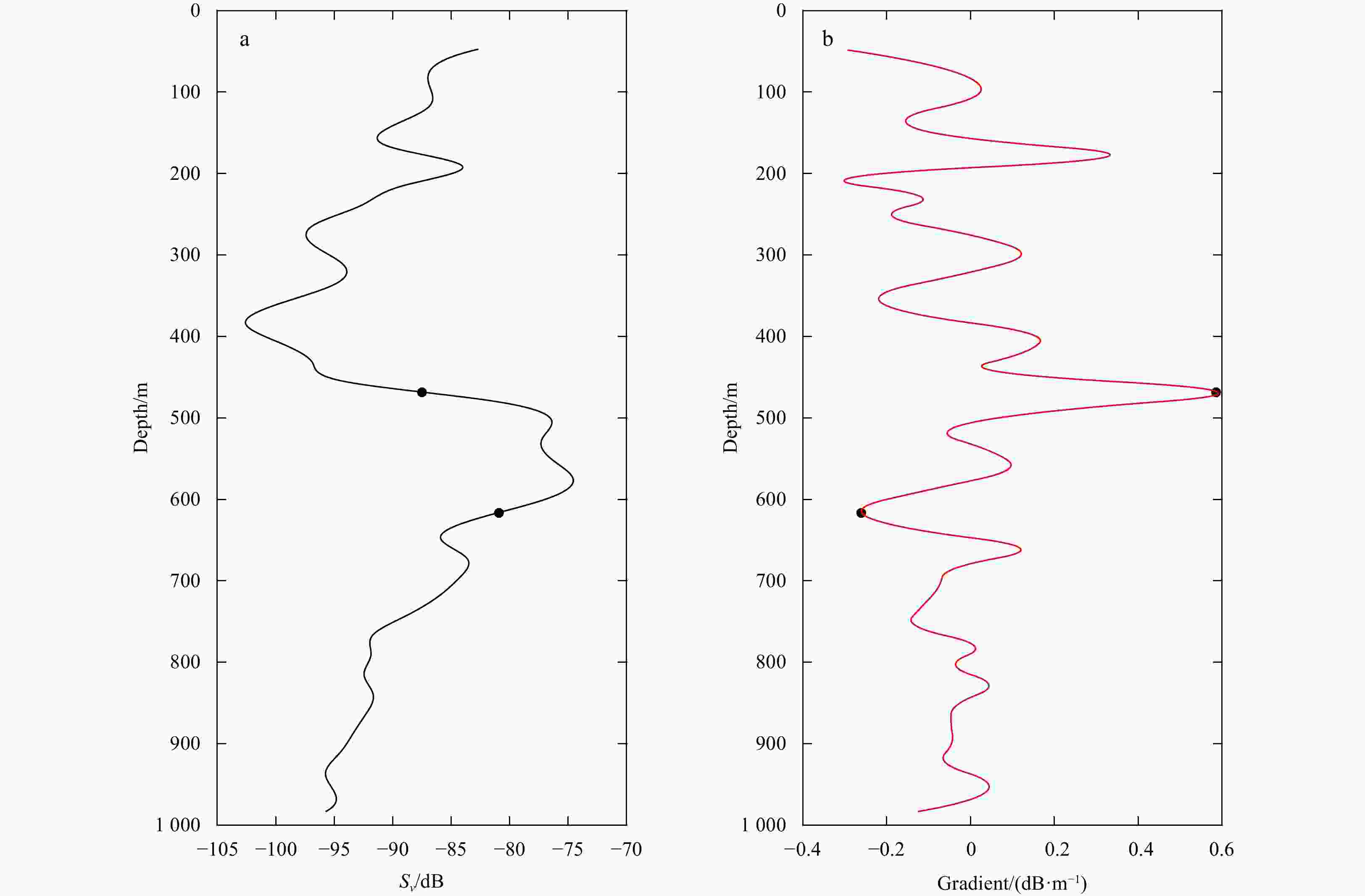
A1. Midday vertical distributions of backscattering strength and its changing gradient in the whole study area. a, b. The vertical distributions of increasing gradient and decreasing gradient, respectively; c. the vertical distribution of mean volume backscattering strength (MVBS). Black curves are identified upper and lower boundaries of DSLs (UBD and LBD). x-axis is the column data number.
Table 1. List of abbreviations in the present study
Acronym Full name Unit NASC (sA) nautical area scattering coefficient m2/n mile2 ASC (sa) area scattering coefficient m2/m2 MGL mesopelagic gathering level % VBC (sv) volume backscattering coefficient m−1 MVBS (Sv) mean volume backscattering strength dB re 4 π m−1 CM center of mass m UBD upper boundary depth m LBD lower boundary depth m MA migration amplitude n mile2/m2 MP migration proportion % WMD weight migration depth m LAC light attenuation coefficient m−1 NPP net primary productivity mg/(m2·d)
(according
to carbon)MLD mixed layer depth m SST sea surface temperature °C SSS sea surface salinity − MAT mesopelagic average temperature °C MAS mesopelagic average salinity − MAO mesopelagic average oxygen concentration μmol/kg Note: − represents no unit. -
[1] Aksnes D L, Røstad A, Kaartvedt S, et al. 2017. Light penetration structures the deep acoustic scattering layers in the global ocean. Science Advances, 3(5): e1602468. doi: 10.1126/sciadv.1602468 [2] Ariza A, Garijo J C, Landeira J M, et al. 2015. Migrant biomass and respiratory carbon flux by zooplankton and micronekton in the subtropical Northeast Atlantic Ocean (Canary Islands). Progress in Oceanography, 134: 330–342. doi: 10.1016/j.pocean.2015.03.003 [3] Béhagle N, Cotté C, Lebourges-Dhaussy A, et al. 2017. Acoustic distribution of discriminated micronektonic organisms from a bi-frequency processing: the case study of eastern Kerguelen oceanic waters. Progress in Oceanography, 156: 276–289. doi: 10.1016/j.pocean.2017.06.004 [4] Béhagle N, Cotté C, Ryan T E, et al. 2016. Acoustic micronektonic distribution is structured by macroscale oceanographic processes across 20–50°S latitudes in the South-Western Indian Ocean. Deep-Sea Research Part I: Oceanographic Research Papers, 110: 20–32. doi: 10.1016/j.dsr.2015.12.007 [5] Benoit-Bird K J, Lawson G L. 2016. Ecological insights from pelagic habitats acquired using active acoustic techniques. Annual Review of Marine Science, 8(1): 463–490. doi: 10.1146/annurev-marine-122414-034001 [6] Bertrand A, Ballón M, Chaigneau A. 2010. Acoustic observation of living organisms reveals the upper limit of the oxygen minimum zone. PLoS ONE, 5(4): e10330. doi: 10.1371/journal.pone.0010330 [7] Bertrand A, Bard F X, Josse E. 2002. Tuna food habits related to the micronekton distribution in French Polynesia. Marine Biology, 140(5): 1023–1037. doi: 10.1007/s00227-001-0776-3 [8] Bianchi D, Galbraith E D, Carozza D A, et al. 2013. Intensification of open-ocean oxygen depletion by vertically migrating animals. Nature Geoscience, 6(7): 545–548. doi: 10.1038/ngeo1837 [9] Bianchi D, Mislan K A S. 2016. Global patterns of diel vertical migration times and velocities from acoustic data. Limnology and Oceanography, 61(1): 353–364. doi: 10.1002/lno.10219 [10] Boswell K M, D’Elia M, Johnston M W, et al. 2020. Oceanographic structure and light levels drive patterns of sound scattering layers in a low-latitude oceanic system. Frontiers in Marine Science, 7: 51. doi: 10.3389/fmars.2020.00051 [11] Brierley A S. 2014. Diel vertical migration. Current Biology, 24(22): R1074–R1076. doi: 10.1016/j.cub.2014.08.054 [12] Carr M E, Friedrichs M A M, Schmeltz M, et al. 2006. A comparison of global estimates of marine primary production from ocean color. Deep-Sea Research Part II: Topical Studies in Oceanography, 53(5−7): 741–770. doi: 10.1016/j.dsr2.2006.01.028 [13] Cascão I, Domokos R, Lammers M O, et al. 2019. Seamount effects on the diel vertical migration and spatial structure of micronekton. Progress in Oceanography, 175: 1–13. doi: 10.1016/j.pocean.2019.03.008 [14] Catul V, Gauns M, Karuppasamy P K. 2011. A review on mesopelagic fishes belonging to family Myctophidae. Reviews in Fish Biology and Fisheries, 21(3): 339–354. doi: 10.1007/s11160-010-9176-4 [15] Cayre P. 1991. Behaviour of yellowfin tuna (Thunnus albacares) and skipjack tuna (Katsuwonus pelarnis) around fish aggregating devices (FADs) in the Comoros Islands as determined by ultrasonic tagging. Aquatic Living Resources, 4(1): 1–12. doi: 10.1051/alr/1991000 [16] Chikuni S. 1985. The fish resources of the Northwest Pacific. Rome: FAO [17] Christiansen B, Denda A, Christiansen S. 2020. Potential effects of deep seabed mining on pelagic and benthopelagic biota. Marine Policy, 114: 103442. doi: 10.1016/j.marpol.2019.02.014 [18] Condie S A, Dunn J R. 2006. Seasonal characteristics of the surface mixed layer in the Australasian region: implications for primary production regimes and biogeography. Marine and Freshwater Research, 57(6): 569–590. doi: 10.1071/MF06009 [19] Coull J R. 1993. World Fisheries Resources. New York: Routledge, 6–18 [20] Deines K L. 1999. Backscatter estimation using broadband acoustic Doppler current profilers. In: Proceedings of the IEEE Sixth Working Conference on Current Measurement. San Diego, CA: IEEE, 249–253 [21] Diogoul N, Brehmer P, Perrot Y, et al. 2020. Fine-scale vertical structure of sound-scattering layers over an east border upwelling system and its relationship to pelagic habitat characteristics. Ocean Science, 16(1): 65–81. doi: 10.5194/os-16-65-2020 [22] Escobar-Flores P, O’Driscoll R L, Montgomery J C. 2013. Acoustic characterization of pelagic fish distribution across the South Pacific Ocean. Marine Ecology Progress Series, 490: 169–183. doi: 10.3354/meps10435 [23] Escobar-Flores P, O’Driscoll R L, Montgomery J C. 2018. Spatial and temporal distribution patterns of acoustic backscatter in the New Zealand sector of the Southern Ocean. Marine Ecology Progress Series, 592: 19–35. doi: 10.3354/meps12489 [24] Everitt B S, Skrondal A. 1998. The Cambridge Dictionary of Statistics. Cambridge: Cambridge University Press, 89 [25] FAO. 2018. The state of world fisheries and aquaculture. Rome: FAO [26] Fennell S, Rose G. 2015. Oceanographic influences on Deep Scattering Layers across the North Atlantic. Deep-Sea Research Part I: Oceanographic Research Papers, 105: 132–141. doi: 10.1016/j.dsr.2015.09.002 [27] Gjøsaeter J, Kawaguchi K. 1980. A review of the world resources of mesopelagic fish. Rome: FAO [28] Godø O R, Samuelsen A, Macaulay G J, et al. 2012. Mesoscale eddies are oases for higher trophic marine life. PLoS ONE, 7(1): e30161. doi: 10.1371/journal.pone.0030161 [29] Gorelova T A. 1984. A quantitative assessment of consumption of zooplankton by epipelagic lanternfishes (family Myctophidae) in the equatorial Pacific Ocean. Journal of Ichthyology, 23: 106–113 [30] Grabowski E, Letelier R M, Laws E A, et al. 2019. Coupling carbon and energy fluxes in the North Pacific Subtropical Gyre. Nature Communications, 10: 1895. doi: 10.1038/s41467-019-09772-z [31] Hernández-León S, Koppelmann R, Fraile-Nuez E, et al. 2020. Large deep-sea zooplankton biomass mirrors primary production in the global ocean. Nature Communications, 11(1): 6048. doi: 10.1038/s41467-020-19875-7 [32] Hu Dunxin, Wu Lixin, Cai Wenju, et al. 2015. Pacific western boundary currents and their roles in climate. Nature, 522(7556): 299–308. doi: 10.1038/nature14504 [33] Ingham M C, Cook S K, Hausknecht K A. 1977. Oxycline characteristics and skipjack tuna distribution in the southeastern tropical Atlantic. Fishery Bulletin, 75(4): 857–865 [34] Inoue R, Kitamura M, Fujiki T. 2016. Diel vertical migration of zooplankton at the S1 biogeochemical mooring revealed from acoustic backscattering strength. Journal of Geophysical Research:Oceans, 121(2): 1031–1050. doi: 10.1002/2015JC011352 [35] Irigoien X, Klevjer T A, Røstad A, et al. 2014. Large mesopelagic fishes biomass and trophic efficiency in the open ocean. Nature Communications, 5: 3271. doi: 10.1038/ncomms4271 [36] Jennings S, Mélin F, Blanchard J L, et al. 2008. Global-scale predictions of community and ecosystem properties from simple ecological theory. Proceedings of the Royal Society B: Biological Sciences, 275(1641): 1375–1383. doi: 10.1098/rspb.2008.0192 [37] Karl D M, Bidigare R R, Letelier R M. 2001. Long-term changes in plankton community structure and productivity in the North Pacific Subtropical Gyre: The domain shift hypothesis. Deep-Sea Research Part II: Topical Studies in Oceanography, 48(8−9): 1449–1470. doi: 10.1016/S0967-0645(00)00149-1 [38] Keeling R F, Körtzinger A, Gruber N. 2010. Ocean deoxygenation in a warming world. Annual Review of Marine Science, 2: 199–229. doi: 10.1146/annurev.marine.010908.163855 [39] Klevjer T A, Irigoien X, Røstad A, et al. 2016. Large scale patterns in vertical distribution and behaviour of mesopelagic scattering layers. Scientific Reports, 6: 19873. doi: 10.1038/srep19873 [40] Klevjer T, Melle W, Knutsen T, et al. 2020. Micronekton biomass distribution, improved estimates across four north Atlantic basins. Deep-Sea Research Part II: Topical Studies in Oceanography, 180: 104691. doi: 10.1016/j.dsr2.2019.104691 [41] Kloser R J, Ryan T E, Young J W, et al. 2009. Acoustic observations of micronekton fish on the scale of an ocean basin: potential and challenges. ICES Journal of Marine Science, 66(6): 998–1006. doi: 10.1093/icesjms/fsp077 [42] Kwong L E, Henschke N, Pakhomov E A, et al. 2020. Mesozooplankton and micronekton active carbon transport in contrasting eddies. Frontiers in Marine Science, 6: 825. doi: 10.3389/fmars.2019.00825 [43] Legendre P, Legendre L. 2012. Numerical Ecology. 3rd ed. Amsterdam: Elsevier, 337–423 [44] Lindstrom M J, Bates D M. 1988. Newton-raphson and EM algorithms for linear mixed-effects models for repeated-measures data. Journal of the American Statistical Association, 83(404): 1014–1022 [45] Lindstrom E, Lukas R, Fine R, et al. 1987. The western equatorial Pacific Ocean circulation study. Nature, 330(6148): 533–537. doi: 10.1038/330533a0 [46] Liu H, Nolla H A, Campbell L. 1997. Prochlorococcus growth rate and contribution to primary production in the equatorial and subtropical North Pacific Ocean. Aquatic Microbial Ecology, 12(1): 39–47 [47] Longhurst A R. 2007. Ecological Geography of the Sea. 2nd ed. London: Academic Press, 327–385 [48] Longhurst A R, Glen Harrison W. 1989. The biological pump: profiles of plankton production and consumption in the upper ocean. Progress in Oceanography, 22(1): 47–123. doi: 10.1016/0079-6611(89)90010-4 [49] MacLennan D N, Fernandes P G, Dalen J. 2002. A consistent approach to definitions and symbols in fisheries acoustics. ICES Journal of Marine Science, 59(2): 365–369. doi: 10.1006/jmsc.2001.1158 [50] McKelvie D S. 1989. Latitudinal variation in aspects of the biology of Cyclothone braueri and C. microdon (Pisces: Gonostomatidae) in the eastern North Atlantic Ocean. Marine Biology, 102(3): 413–424. doi: 10.1007/BF00428494 [51] Miller K A, Thompson K F, Johnston P, et al. 2018. An overview of seabed mining including the current state of development, environmental impacts, and knowledge gaps. Frontiers in Marine Science, 4: 418. doi: 10.3389/fmars.2017.00418 [52] Moline M A, Benoit-Bird K, O’Gorman D, et al. 2015. Integration of scientific echo sounders with an adaptable autonomous vehicle to extend our understanding of animals from the surface to the bathypelagic. Journal of Atmospheric and Oceanic Technology, 32(11): 2173–2186. doi: 10.1175/JTECH-D-15-0035.1 [53] Mote P W, Salathé E P. 2010. Future climate in the Pacific Northwest. Climatic Change, 102(1−2): 29–50. doi: 10.1007/s10584-010-9848-z [54] Mullison J. 2017. Backscatter estimation using broadband acoustic Doppler current profilers-updated. In: Hydraulic Measurements & Experimental Methods Conference. Durham, NH: ASCE [55] Netburn A N, Anthony Koslow J. 2015. Dissolved oxygen as a constraint on daytime deep scattering layer depth in the southern California current ecosystem. Deep-Sea Research Part I: Oceanographic Research Papers, 104: 149–158. doi: 10.1016/j.dsr.2015.06.006 [56] Oestreich W K, Ganju N K, Pohlman J W, et al. 2016. Colored dissolved organic matter in shallow estuaries: relationships between carbon sources and light attenuation. Biogeosciences, 13(2): 583–595. doi: 10.5194/bg-13-583-2016 [57] Padial A A, Thomaz S M. 2008. Prediction of the light attenuation coefficient through the Secchi disk depth: empirical modeling in two large Neotropical ecosystems. Limnology, 9(2): 143–151. doi: 10.1007/s10201-008-0246-4 [58] Pakhomov E A, Podeswa Y, Hunt B P V, et al. 2019. Vertical distribution and active carbon transport by pelagic decapods in the North Pacific Subtropical Gyre. ICES Journal of Marine Science, 76(3): 702–717. doi: 10.1093/icesjms/fsy134 [59] Phillips A J, Brodeur R D, Suntsov A V. 2009. Micronekton community structure in the epipelagic zone of the northern California Current upwelling system. Progress in Oceanography, 80(1−2): 74–92. doi: 10.1016/j.pocean.2008.12.001 [60] Polis G A, Anderson W B, Holt R D. 1997. Toward an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annual Review of Ecology and Systematics, 28(1): 289–316. doi: 10.1146/annurev.ecolsys.28.1.289 [61] Prince E D, Goodyear C P. 2006. Hypoxia-based habitat compression of tropical pelagic fishes. Fisheries Oceanography, 15(6): 451–464. doi: 10.1111/j.1365-2419.2005.00393.x [62] Proud R, Cox M J, Brierley A S. 2017. Biogeography of the global ocean’s mesopelagic zone. Current Biology, 27(1): 113–119. doi: 10.1016/j.cub.2016.11.003 [63] Receveur A, Kestenare E, Allain V, et al. 2020. Micronekton distribution in the Southwest Pacific (New Caledonia) inferred from shipboard-ADCP backscatter data. Deep-Sea Research Part I: Oceanographic Research Papers, 159: 103237. doi: 10.1016/j.dsr.2020.103237 [64] Salvanes A G V, Kristoffersen J B. 2001. Mesopelagic fishes. In: Steel J, Thorpe S, Turekian K, eds. Encyclopedia of Ocean Sciences. San Diego: Academic Press, 1711–1717 [65] Sato M, Benoit-Bird K J. 2017. Spatial variability of deep scattering layers shapes the Bahamian mesopelagic ecosystem. Marine Ecology Progress Series, 580: 69–82. doi: 10.3354/meps12295 [66] Seibel B A. 2011. Critical oxygen levels and metabolic suppression in oceanic oxygen minimum zones. Journal of Experimental Biology, 214(2): 326–336. doi: 10.1242/jeb.049171 [67] Shen G, Shi B. 2002. Primary Production in Marine. Beijing: Science Press, 189–223 [68] Sibson R. 1981. A brief description of natural neighbor interpolation. In: Barnett V, ed. Interpreting Multivariate Data. New York: John Wiley & Sons, 21–36 [69] Smeti H, Pagano M, Menkes C, et al. 2015. Spatial and temporal variability of zooplankton off New Caledonia (Southwestern Pacific) from acoustics and net measurements. Journal of Geophysical Research: Oceans, 120(4): 2676–2700. doi: 10.1002/2014JC010441 [70] St John M A, Borja A, Chust G, et al. 2016. A dark hole in our understanding of marine ecosystems and their services: Perspectives from the mesopelagic community. Frontiers in Marine Science, 3: 31 [71] Stedmon C A, Nelson N B. 2015. The optical properties of DOM in the ocean. In: Hansell D A, Carlson C A, eds. Biogeochemistry of Marine Dissolved Organic Matter. 2nd ed. San Diego: Academic Press, 481–508 [72] Steinacher M, Joos F, Frölicher T L, et al. 2010. Projected 21st century decrease in marine productivity: a multi-model analysis. Biogeosciences, 7(3): 979–1005. doi: 10.5194/bg-7-979-2010 [73] Steinberg D K, Cope J S, Wilson S E, et al. 2008a. A comparison of mesopelagic mesozooplankton community structure in the subtropical and subarctic North Pacific Ocean. Deep-Sea Research Part II: Topical Studies in Oceanography, 55(14−15): 1615–1635. doi: 10.1016/j.dsr2.2008.04.025 [74] Steinberg D K, Van Mooy B A S, Buesseler K O, et al. 2008b. Bacterial vs. zooplankton control of sinking particle flux in the ocean’s twilight zone. Limnology and Oceanography, 53(4): 1327–1338. doi: 10.4319/lo.2008.53.4.1327 [75] Stramma L, Prince E D, Schmidtko S, et al. 2012. Expansion of oxygen minimum zones may reduce available habitat for tropical pelagic fishes. Nature Climate Change, 2(1): 33–37. doi: 10.1038/nclimate1304 [76] Toole J M, Zou E, Millard R C. 1988. On the circulation of the upper waters in the western equatorial Pacific Ocean. Deep-Sea Research Part A. Oceanographic Research Papers, 35(9): 1451–1482 [77] Urmy S S, Horne J K. 2016. Multi-scale responses of scattering layers to environmental variability in Monterey Bay, California. Deep-Sea Research Part I: Oceanographic Research Papers, 113: 22–32. doi: 10.1016/j.dsr.2016.04.004 [78] Urmy S S, Horne J K, Barbee D H. 2012. Measuring the vertical distributional variability of pelagic fauna in Monterey Bay. ICES Journal of Marine Science, 69(2): 184–196. doi: 10.1093/icesjms/fsr205 [79] Zhang Dongsheng, Wang Chunsheng, Liu Zhensheng, et al. 2012. Spatial and temporal variability and size fractionation of chlorophyll a in the tropical and subtropical Pacific Ocean. Acta Oceanologica Sinica, 31(3): 120–131. doi: 10.1007/s13131-012-0212-1 -




 下载:
下载: